The Leukemia & Lymphoma Society (LLS) Therapy Acceleration Program® (TAP)
WHAT IS TAP?
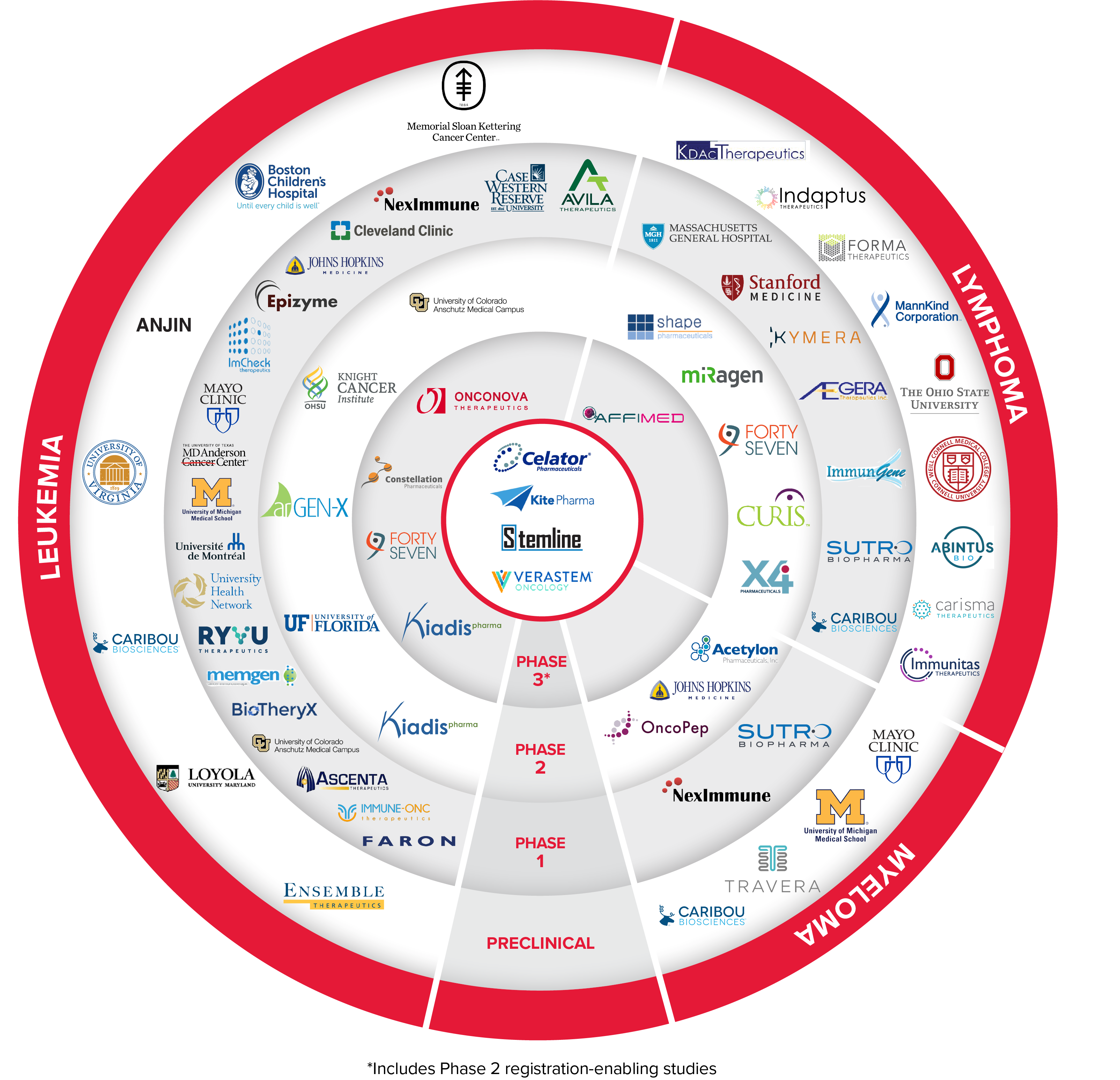
TAP is a strategic venture philanthropy funding initiative to accelerate high-risk, innovative blood cancer therapeutics and change the standard of care in leukemia, lymphoma, and multiple myeloma.
Since establishment in 2007, TAP has invested >$130 million in over 70 projects.
Four TAP-supported therapies have been approved by the U.S. Food and Drug Administration (FDA) or included in the National Comprehensive Cancer Network (NCCN) Guidelines:
- CPX-351 (Vyxeos®), first approved treatment (an innovative reformulation of two chemotherapies) for patients with certain types of high-risk acute myeloid leukemia (AML)
- Axicabtagene ciloleucel (Yescarta®), first CAR T-cell immunotherapy approved for patients with non-Hodgkin lymphoma (NHL) and transformed follicular lymphoma (tFL)
- Tagraxofusp-erzs (Elzonris®), first approved therapy for children and adults with blastic plasmacytoid dendritic cell neoplasm (BPDCN)
- Duvelisib (Copiktra®), first dual inhibitor of PI3K-delta and gamma pathways included in NCCN Guidelines for patients with all subtypes of peripheral T-cell lymphoma (PTCL)
Currently, there are over 20 TAP-supported companies with assets in active development, including 4 ongoing registration-enabling clinical studies in blood cancer & 2 beyond blood cancer.
To learn more about TAP: https://www.lls.org/tap
LLS leverages its over 70-year commitment to cures for blood cancers and has funded over 4000 academic grants and 70 TAP projects since its inception, amounting to $1.3 Billion of support toward disease control and cures. LLS knowledge and know-how in blood cancer drug development is unparalleled, underscored by LLS support for over 85% of the new blood cancer therapies approved in the past 20 years.
To learn more about LLS-supported Research: https://www.lls.org/lls-funded-research